 
If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. 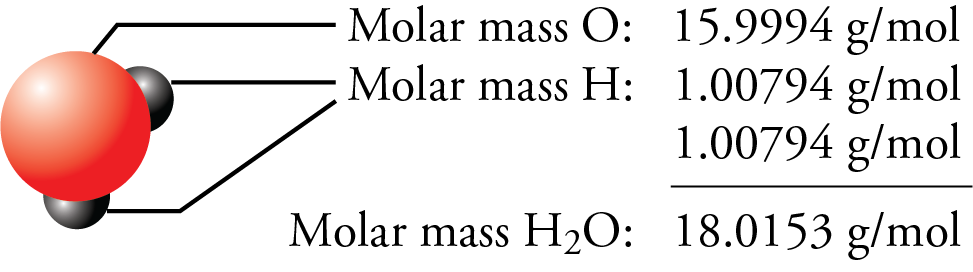
Table 3.4.1 Comparison of the properties predicted by Mendeleev in 1869 for eka-aluminum and eka-silicon with the properties of gallium (discovered in 1875) and germanium (discovered in 1886) Property The observed properties of gallium and germanium matched those of eka-aluminum and eka-silicon so well that once they were discovered, Mendeleev’s periodic table rapidly gained acceptance. Two of the blanks Mendeleev had left in his original table were below aluminum and silicon, awaiting the discovery of two as-yet-unknown elements, eka-aluminum and eka-silicon (from the Sanskrit eka, meaning “one,” as in “one beyond aluminum”). The most convincing evidence in support of Mendeleev’s arrangement of the elements was the discovery of two previously unknown elements whose properties closely corresponded with his predictions ( Table 3.4.1 ). Until roughly 1960, a rectangular table developed from Mendeleev's table and based on reactivity was standard at the front of chemistry lecture halls. Notice how this approach has trouble with the transition metals. In Group VII, one atom of fluorine (F), chlorine (Cl), and bromine (Br), reacts with one atom of hydrogen. For example, in Group I, two atoms of hydrogen (H), lithium (Li), sodium (Na), and potassium (K) form compounds with one atom of oxygen. The groups in Mendeleev's table are determined by how many oxygen or hydrogen atoms are needed to form compounds with each element. Atomic masses are shown after equal signs and increase across each row from left to right. In Meyer’s plot of atomic volume versus atomic mass, the nonmetals occur on the rising portion of the graph, and metals occur at the peaks, in the valleys, and on the down slopes.įigure 3.4.3 Mendeleev’s periodic table, as published in the German journal Annalen der Chemie und Pharmacie in 1872: The column headings “Reihen” and “Gruppe” are German for “row” and “group.” Formulas indicate the type of compounds formed by each group, with “R” standing for “any element” and superscripts used where we now use subscripts. Trending Questions What is the symbol equation for chlorine and carbon combined? What types of scientific instruments are found in a chemistry class room? What gase is used in advertising signs? What is dangerous creatinine level? When did Purex bleach stop coming in glass bottles? What three elements are in lithium sulfate? M8 and M9 paper emit toxic fumes when burned true or false? A blended mixture of two or more solids liquids and gases is called what? What is a noninvasive procedure used to measure the oxygen level of a patient's blood? What is scrap price of 23k gold per gram? What is the number of CO2 molecules in 0.\( \newcommand\)Īs shown in Figure 3.4.2, the alkali metals have the highest molar volumes of the solid elements. Calculate the percentage of water in hydrate.Divide the molar mass of water by the molar mass of the hydrate, and multiply result by 100%.36.04g147.01g x 100%Percent water in hydrate is 24.52%. This number may be useful to remember on the day of the test or while doing practice problems.*2. Add these values together to find the molar mass of the hydrate.Molar Mass Anhydrate + Molar Mass Water Molecules* = Molar Mass Hydrate* Tip: the molar mass of water for all hydrate calculations is 18.02g x number of water molecules. Do this for both the anhydrate and the water molecules. etc.Add up all the mass values and you have the value for molar mass. Find the molar mass of the hydrate (Calcium Chloride Dihydrate).Find the molar mass of water and the anhydrate (anhydrate + water = hydrate) add the molar mass values of each to find the molar mass of the hydrate.Molar Mass CaCl2: 110.98g+ Molar Mass H2O: 36.04g*Molar Mass CaCl2 * 2H2O: 147.01gFinding Molar Mass# atoms element A * atomic mass element A = Mass A# atoms element B * atomic mass element B = Mass B. What is the theoretical percentage of water of hydration in cacl2-2h20?ġ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
